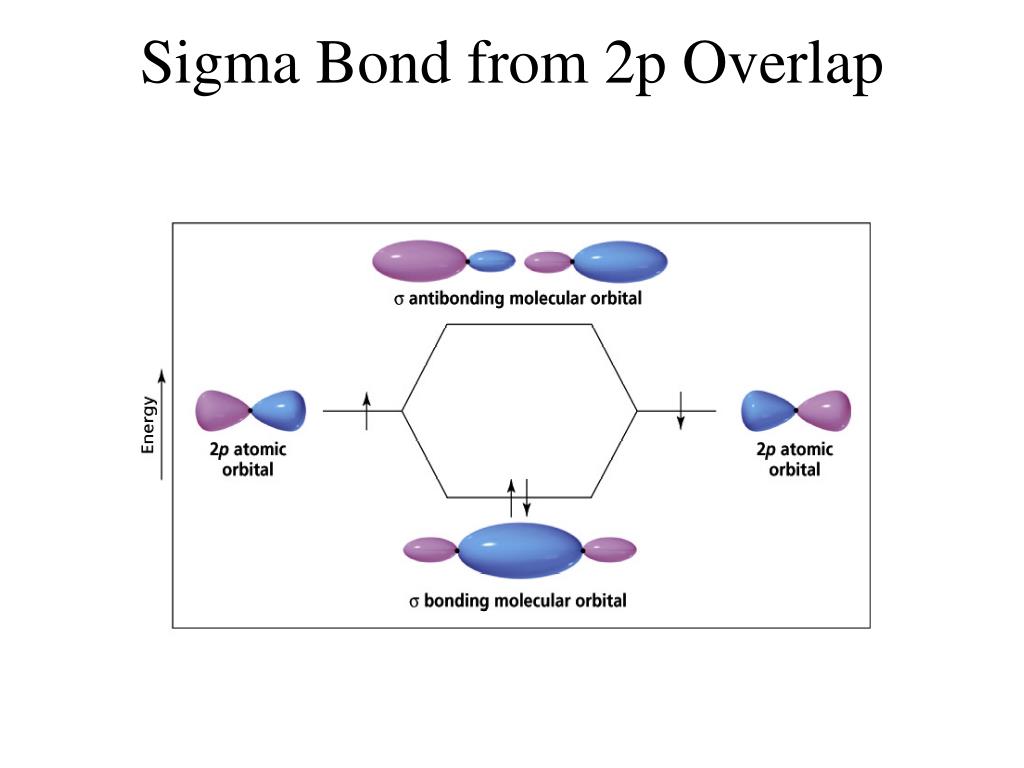
A double bond contains a one sigma bond and one Pi bond eg. In the case of multiple bonds like a triple and double bond, it contains pi bonds too. A single bond always contains only one sigma bond and no pi bond is there eg. In the case of a Pi bond which is formed due to the lateral overlapping of orbitals and the bond is weaker than a sigma bond. Sigma bond is usually defined for diatomic molecules and is formed by the headways overlapping between atomic orbitals. 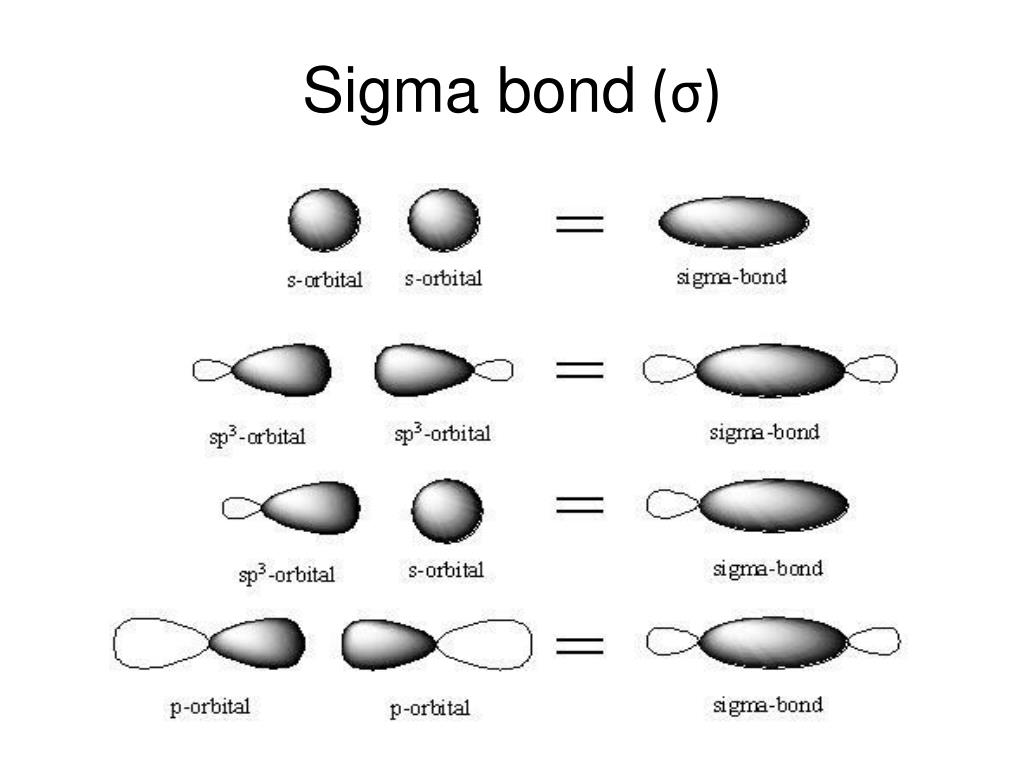
The following image shows the pi bond formation. The following images show the formation of a sigma bond between two atoms.įor a pi bond formation, the atomic orbitals are overlapped in a sideways manner. The orbital overlapping of sigma and pi bonds is different, for a sigma bond formation headways overlapping between atomic orbitals will take place. More than one pi bond can be formed between atoms.ĭistinguishing features of sigma and pi bonds: 
The sigma bond that can be formed between two atoms is one. Overlapping orbitals are only pure orbitals. The overlapped orbitals formed by pure orbitals, hybrid orbitals and one hybrid orbital, and one pure orbital. It will determine the shape of the molecule.ĭo not have a role in the shape of molecules. Pi bonds can be accessed only with the help of a sigma bond. It can exist independently and free rotation is possible. Pi bonds are formed by the lateral overlapping of atomic orbitals. Sigma bonds are formed by the headways overlapping of atomic orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
